|
This radius is noticed to differ between 98 pm to 108 pm depending one the different circumstances. Chlorine ionic/covalent radiusĬhlorine’s covalent radius has been identified as approximately 102pm. This radius is denoted by r w and it refers to the half of the distance between two single atom of Chlorine is 175 pm within a balance electrostatic force. Chlorine Van der Waals radiusĬhlorine has 175 pm of van der Waals radius. This boiling point is also noticed to be lower than other elements but higher than Fluorine. Boiling point of Chlorine has been recognised as −34.04 ☌ (239.11 K, −29.27 ☏). It can be said that the meting point of Cl is very low but higher than the same of Fluorine. The melting point of Chlorine is noticed to be −101.5 ☌ (171.6 K, −150.7 ☏). At STP, standard amount of atomic density is 3.2 g/L. The atomic density of Chlorine has been obtained as 3.214g/cm3. In Pauling scale of electronegativity, the electronegativity of Chlorine has been measures by 3.16. Chlorine Electronegativity according to Pauling Chlorine atomic number Chlorine atomic WeightĪtomic weight of Chlorine has been calculated as approximately 35.453 u. This means that Chlorine has 17 protons and 17 electrons in a single atom. Chlorine atomic numberĬhlorine is noticed to have atomic number of 17. 
Chlorine block in periodic tableĬhlorine has been placed at d-block by Mendeleev, who was the former of the modern periodic table. This period of selection of Chlorine has been done by the number of electronic shell present in its atom. Chlorine period in periodic tableĬhlorine can be identified in period 2 in the periodic table. Therefore, it can be said that groups are the columns in periodic table. Chlorine group in periodic tableĬhlorine is placed at group 17 where all the other Halogen elements are placed vertically. As ‘C’ and ‘l’ are emphasised in the word ‘Chlorine’, these two letters is used as its symbol. “Cl” is used as symbol of Chlorine in chemistry to represent it in chemical reactions. All the relevant facts such as meting point, boiling point, electronegativity, ionisation energies and others will be discussed throughout this article. The chemical facts of the element are reliable in keenly examining the internal facts and chemical behaviour of element in chemical reaction with others.Ĭhemical properties of Chlorine including its position in the periodic table would be described in this article. It is the second halogen element in the periodic table. Let us continue describing the chemical properties of Chlorine below.Ĭhlorine chemical properties at first include the behaviour of Chlorine elements as a halogen. Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms.Chlorine chemical properties cover some relevant chemical facts about the periodic element Chlorine. For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. 
Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. 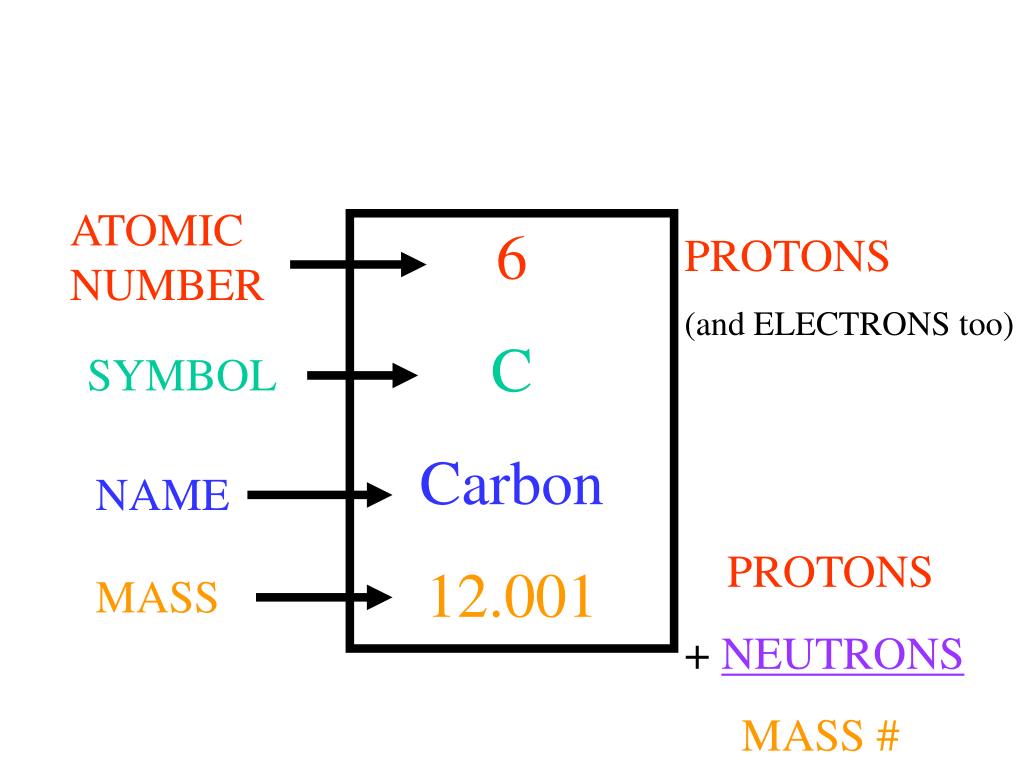
The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
